|
Many laboratory protocols require the serial dilution of reagents or compounds. IC50 assays, commonly used to evaluate drug efficacy, and assay development procedures, as well as standard-curve generation, involve the serial dilution of compounds, proteins, or detection agents. These processes can be streamlined by utilizing automated liquid-handling equipment with serial dilution capabilities. Serial dilution processes face two major challenges.
The first is error propagation across columns or rows. With each sequential serial dilution step, transfer inaccuracies lead to less accurate and less precise dispensing. The result is that the highest dilutions will have the most inaccurate results.
To compensate for this error possibility, longer mixing times are required, which then increases the time required to perform the serial dilution. These challenges greatly limit the throughput capacity of an automated serial dilution system. To overcome these challenges, the effects of various mixing parameters of a serial dilution protocol were explored. Velocity11’s (www.velocity11.com) Bravo™ Liquid Handling Platform performed serial dilution with the same pipette head as a full plate dispenser (Figure 1).
With the platform’s VWorks™ software, the application allowed the total control of liquid transfer and mixing heights and speeds, which allowed efficient exploration of mixing parameters. The goals were to determine which parameters had the greatest effect on mixing and to reduce the time required to perform a serial dilution. Serial Dilution Mix Cycles The basic experiment diluted fluorescein across the columns of a 96-well plate, from A1 to A10 (A11 and A12 were blank wells).
The starting volume was 300 µL, and 200 µL tips were utilized for the transfer (150 µL, a 1:2 dilution) and mixing steps (190 µL). There are two main components of an accurate and precise serial dilution: the accuracy and precision of the transfer and the efficiency of mixing. Transfers were previously determined to have a precision and accuracy of 99% at this volume; any observed deviations in precision and accuracy were due to error propagation from ineffective mixing. Two measures were used to evaluate mixing efficiency.
The Coefficient of Variance (CV) of each column indicated the precision of the mixing step. The CV also provided information on the propagation of error across a plate—the CV increased sharply across the plate if mixing was incomplete. The second indication was the accuracy of the transfer. A calibration curve was prepared, and each experimental dilution concentration was plotted against the standard curve to determine the real concentrations in each column. The first experiment varied the number of mixing cycles between 3 and 20. The average precision (averaging CVs for columns 1–10) improved asymptotically as the number of mix cycles increased. Three mixes before each transfer yielded an average CV of 11.8%, while 20 mixes gave a considerably better CV of 1.7%.
The precision in all cases generally worsened as the serial dilution proceeded across the plate; this was expected as the error in the earlier columns propagated with each transfer. The accuracy ratio improved as the number of mix cycles increased. The accuracy ratio is an average of the concentration of the diluted column compared to the previous column—a perfect serial dilution has an accuracy ratio of 1:2.00 across the entire plate. The accuracy ratio of the plate improved with more mix cycles, improving from 1:1.85 to 1:2.01.
While the precision and accuracy with 20 mix cycles is close to a perfect serial dilution, the length of time required might be considered impractical. The 20-mix cycle protocol required 20 minutes per plate, while a three-mix cycle protocol required less than six minutes. Efforts were then focused on the factors that could improve the three-mix cycle protocol to produce accuracy and precision results consistent with the 20-mix cycle protocol. Mix Tip Height The mix tip height was modified in order to determine the effect of distributing the liquid at different locations in the well.
As the mix tip height was raised, the average precision improved. At a height of 3 mm from the bottom of the well, the average precision was 3.9%.
The precision worsened as the tip distance from the bottom of the well decreased, reaching a CV of 15% at a height of 0.1 mm. Accuracy tracked with precision, and the higher mix height also improved the accuracy ratio to 1.95. This trend is possibly because the higher dispense height ensures that more of the sample was circulated by the mix cycle. In a mix roughly in the middle of the well volume, dispensed liquid is forced toward the well bottom while dispensing, and aspirated liquid is pulled from the center of the well. If the mix occurs close to the bottom of the plate, the dispensed liquid is pulled back into the tip during the aspiration. Mixing in the center allows the dispensed liquid to be more evenly distributed in the sample, thus increasing the likelihood of efficient mixing. Mix Liquid Class Setting The VWorks software controlling the Bravo platform allows the creation of liquid classes, which allows the operator to modify the velocity and acceleration for aspirating, dispensing, and mixing tasks.
The original liquid class settings for the mix were 100 µL/s velocity and 500 µL/s2 acceleration. Precision and accuracy improved as the mix velocity increased.
This effect plateaus; above 300 µL/s, there is no appreciable improvement in increasing the speed. The cause of this is likely due to the creation of more turbulent mixing, which in turn distributed the fluorescein dye more quickly throughout the solution. Dynamic Tip Retraction/Extension Finally, the effect of dynamic tip retraction and extension was explored. This function moved the tips deeper into the well during each aspirate step, and retracted them during each dispense step. This allowed a larger volume of the well to be effected by the mix step by adding the movement of the tip into the mix task.
There was a marginal improvement (less than 0.5% improvement in CV/accuracy) observed in using this technique. Additionally, no effect was observed by utilizing another mix standard, which involved aspirating close to the bottom of the well and dispensing near the top of the solution.
This mixing method caused no improvement once the other parameters described above had been optimized. These experiments mixed homogenous solutions; there may be an improvement with this technique if the solutions are expected to have different viscosities. Based on these experiments, the parameters that had the largest impact on efficient mixing were (in decreasing order):. Speed of the mixing step.
Height of the tip during the mix. Tip-retraction capabilities To verify this conclusion, the first experiment (varying the number of mix cycles) was repeated with the improved mix parameters. The new parameters provided increased precision and accuracy, and improved the accuracy and precision of the 3-mix cycle operation to a level comparable with the 20-mix cycle operation (Figure 2). More importantly, the new parameters also decreased the time required to run an effective serial dilution protocol from 20 minutes to just under 5 minutes. This has tremendous potential in automating a serial dilution assay and ensuring accurate and precise results.
In vitro evolution of RNA molecules requires a method for executing many consecutive serial dilutions. To solve this problem, a microfluidic circuit has been fabricated in a three-layer glass-PDMS-glass device. The 400-nL serial dilution circuit contains five integrated membrane valves: three two-way valves arranged in a loop to drive cyclic mixing of the diluent and carryover, and two bus valves to control fluidic access to the circuit through input and output channels. By varying the valve placement in the circuit, carryover fractions from 0.04 to 0.2 were obtained. Each dilution process, which is comprised of a diluent flush cycle followed by a mixing cycle, is carried out with no pipeting, and a sample volume of 400 nL is sufficient for conducting an arbitrary number of serial dilutions.
Mixing is precisely controlled by changing the cyclic pumping rate, with a minimum mixing time of 22 s. This microfluidic circuit is generally applicable for integrating automated serial dilution and sample preparation in almost any microfluidic architecture. INTRODUCTION Serial dilution is among the most fundamental and widely practiced laboratory techniques, with applications ranging from measuring detector response, to determining kinetic rate constants, to culturing cells. Serial dilution is particularly important in directed evolution experiments in which a population of RNA molecules is made to undergo repeated rounds of selective amplification.
In order to evolve molecules with interesting properties, it is necessary to propagate the population of RNAs through many logs of selective growth. This is accomplished by serially diluting an aliquot of the reaction mixture into fresh growth medium at regular intervals. Performing serial dilutions by manual pipeting is a mundane and time-consuming task that has limited the execution of highly longitudinal experiments in molecular evolution. Microfluidic technology presents a practical solution to this problem by automating the fluid handling associated with serial dilution.
The core strengths of microfluidic technology are integration, high throughput, and low-volume handling. Microfluidic analogs outperform conventional instrumentation with regard to speed, throughput, and reagent consumption by an order of magnitude or more, and allow integration of sample preparation and analysis in a single device., Precise manipulation of fluids in these devices is achieved by electrokinetic control, - microfabricated membrane valves, or various other approaches to microfluidic transport and control. The combination of highly ordered flow and precise manipulation allows one to carry out diverse synthetic and analytical methods with remarkable control., Despite the near universal need for the preparation of standard samples, little work has been done to miniaturize and to expedite this process. Approaches have included variously configured splitter channels - and differential metering of multiple inputs into addressable microfabricated assay wells. Each of these approaches to serial dilution requires N independent outputs (splitter branches, end reactors, etc.) for N consecutive dilutions, making them unsuitable for executing an arbitrary number of dilutions. The ideal circuit would automate sample and diluent metering and mixing, while scaling to an arbitrary number of serial dilutions.
A microfluidic mixing loop addresses mixing requirements by reducing effective diffusion lengths, while providing a compact geometry for manipulating nanoliter volumes. A microfluidic serial dilution circuit that implements these advantageous mixing and scaling characteristics and incorporates sample metering elements has been designed, fabricated, and characterized. It is compact and does not geometrically constrain the number of possible serial dilutions. Precise metering of the sample carryover fraction and rapid, reproducible mixing of the diluent with the carryover are achieved in the same structure. The device is computer controlled, and the preparation of successive serial dilutions is fully automated.
Because the circuit employs microfluidic pumping, serially diluted sample aliquots can easily be routed from the dilution circuit to other microfluidic components, such as a separation channel or microreactor. Microdevice Fabrication and Design The three-layer glass-PDMS-glass sandwich structure was fabricated as described previously., Features on the fluidic and manifold glass wafer layers were isotropically etched to a depth of 50 μm. The etched fluidic and manifold layers were visually aligned and reversibly bonded to one another with an intervening optically transparent PDMS membrane (250 μm thick, Rogers Corporation, Carol Stream, IL). Visual alignment and reversible bonding was performed in a laminar flow hood to minimize particulate contamination of the clean glass wafers and PDMS membrane.
Nylon tubing barbs (1/16″) were affixed to the fluidic chip surface at five pneumatic access holes to interface pneumatic control line tubing with the device. All reservoirs and vacuum access holes were drilled with 1.1-mm-diameter diamond-coated drill bits. A schematic of the microfluidic serial dilution circuit is shown in. Fluidic channels (black) are 300 μm wide, and valve deflection chambers (gray) are 1 mm in diameter.
Both layers are 50 μm deep. All dimensions are after isotropic etching. Two-way valves A, B, and C control fluid flow in the loop. Bus valves I and O control fluidic access to the input and output reservoirs, R i and R o, respectively. The loop remains continuous when the bus valves are closed, but fluid flow from R i and to R o is prevented. The boxed inset to depicts the cross-sectional view of the glass-PDMS-glass sandwich structure. Schematic of the microfluidic serial dilution circuit.
Fluidic channels are shown in black and pneumatic features are shown in gray. The input and output fluidic access reservoirs (1.1-mm diameter) are labeled R i and R o, respectively. The five membrane valve deflection chambers are labeled A, B, C, I, and O on their respective pneumatic lines. Valves A, B, and C are two-way valves and are continuous only when open. Input and output valves I and O are bus valves, connecting R i and R o to the mixing loop.
When open, I and O allow flow from R i and R o to and from the mixing loop. Fluidic continuity is preserved within the mixing loop even when I and O are closed. The boxed diagram depicts a cross section of the device at a two-way valve junction, showing the fluidic and manifold wafers, the PDMS membrane, the fluidic channel and discontinuity, and the corresponding valve displacement chamber. Pneumatic Control Computer-controlled pneumatic actuation of the membrane valves was accomplished using a TTL-driven vacuum solenoid valve array (HV010, Humphrey, Kalamazoo, MI).
On TTL low, the solenoid directs atmospheric pressure output, and the associated membrane valve rests in the closed state. On TTL high, the solenoid switches to vacuum and causes the associated membrane valve to deflect open. The solenoid array is driven by the digital output of a NI6715 data acquisition PCMCIA card and PC laptop with software written in house (LabVIEW, National Instruments, Austin, TX). A sequence of valve states defines a pumping program. A variable hold step interposed between states in the sequence is the valve actuation time. Three pumping programs were written to manipulate fluid in the serial dilution circuit. The valve sequences of each pumping program are written showing only the open valves at each step, and the hold step is indicated by a comma after each state in the sequence.
For example, the program (AB, B) starts with valves A and B open and valves C, I, and O closed. This state is followed by a hold step, then valve A is closed leaving only B open. The mix pumping program is the valve state sequence (A, AB, B, BC, C, AC).
The flush pumping program is the valve state sequence (A, AB, B, BO, IO, IA). The prime pumping program is the valve state sequence (I, ACI, AC, ABCO, BO, O). Looping a pumping program results in continuous pumping., Each pumping program requires two input parameters for operation: the valve actuation time (in milliseconds) and the length of time the program is iterated (in seconds).
Fluidic manipulation protocols are described in the text using the format: program(valve actuation time,iteration time), with valve actuation times given in milliseconds and iteration times given in seconds. For example, mix(80,60) indicates that the mix program is run with 80 ms valve actuation time, iterated for 60 s. Flow Visualization and Device Characterization Flow in the channels was visualized using a solution of fluorescein dye (10 μM in TAE) and a fiber-coupled epifluorescence microscope (488-nm laser excitation), which has been described. Epifluorescence movies of the various pumping programs were acquired using a 12-bit CoolSnap FX CCD (10 fps, 50-ms exposure, 8 × 8 pixel binning, Roper Scientific, Tucson, AZ). The illumination area was ∼1.2 cm diameter and the power density was 1 mW/mm 2.
Confocal fluorescence data were acquired using an inverted microscope fabricated in house. Laser excitation from a frequency-doubled diode laser was coupled into the optical detection train with a dichroic long-pass mirror (505DRLP, Omega Optical, Brattleboro, VT) and focused on the microfluidic channels with an infinite conjugate microscope objective (40× 0.6 NA, Newport, Irvine, CA). Fluorescence was collected with the same objective, spectrally filtered with a bandpass filter (535DF60, Omega Optical), and focused with a 100-mm focal length achromatic lens on a 100- μm pinhole before impinging a photon counting PMT (H7827, Hamamatsu Corp., Japan). For all confocal fluorescence measurements, the detector was positioned in the fluidic channel region bounded by valves A and B. Fluid handling characteristics of the device were quantitated using confocal fluorescence microscopy. The input reservoir, R i, was spotted with fluorescein solution and the circuit was run with prime(200,30) to prime with dye. A syringe loaded with TAE buffer (the diluent) was used to rinse away residual dye solution in R i and to load diluent.
This standard procedure was used to prepare the circuit for each of the following device characterization studies. The intrinsic carryover fraction (CF) for each serial dilution circuit was determined.
The average fluorescence signal of the concentrated dye was measured, then the circuit was run with flush(100,60), and the average buffer background fluorescence signal was measured. Finally, the circuit was run with mix(100,120) to mix the carryover into the diluent. After mixing, the average fluorescence signal of the diluted dye was measured. The ratio of the background-subtracted diluted dye signal to the dye concentrate signal is the CF. To demonstrate multiple serial dilutions of the same sample, a sample of 10 μM fluorescein was diluted in TAE using a mixing loop with CF of 0.2.
To increase dynamic range, an OD 1 neutral density filter (Newport) was placed in line to measure the sample concentrate fluorescence intensity. Thereafter, the filter was removed and the fluorescence intensity of each consecutive dilution was measured as described above. Fluidic handling reproducibility was evaluated by performing replicate dilutions. For each replicate, the circuit was prepared as described. Then the circuit was run with flush(100,90), followed by mix(100,120).
Mixing was characterized by performing dilutions with variable valve actuation time during the mixing step. The circuit was primed as described, and mix(x,500) was initiated, where x was systematically varied from 300 ms to 50 ms. RESULTS AND DISCUSSION The serial dilution of an analyte can be automated and carried out on the nanoliter scale using an appropriately configured microfluidic mixing loop. In-line computer-controlled membrane valves allow precise fluidic manipulation, automation, and parallelization. Fluidic operations, such as diluent flushing, mixing, and priming can be accurately and precisely performed without manual intervention, and performed simultaneously in many parallel circuits.
A quantitative description of device performance was developed using epifluorescence flow visualization and confocal fluorescence microscopy. Epifluorescence visualization of the pumping programs flush and mix is presented in. Diluent is pumped into the circuit through I, then through A and B, and finally out of the circuit through O. A plug of material in the region bounded by valves I and O and containing C is preserved by flush. Frames 1 through 4 show TAE buffer (diluent) being pumping from R i to R o around the right side of the fluorescein dye-primed circuit. A plug of fluorescein dye (carryover) is preserved on the left (frame 4).
The carryover and diluent are mixed together in the mix operation by serially actuating valves A, B, and C while keeping valves I and O closed. Frames 5 through 8 show the carryover being mixed into the diluent as the fluid is cyclically pumped, and the fluorescence intensity in the loop homogenizes. Serial dilution circuit pumping program schematics and epifluorescence stills. Still frames are 50-ms exposures. The circuit is initially primed with fluorescein dye. Fluid flow paths are indicated with gray arrows overlaid on the circuit schematic.
The flush program is used for diluent flushing and carryover isolation, and is accomplished by serially actuating I, A, B, and O while keeping C closed. Buffer is pumped from R i to R o, clearing the right side of the mixing loop while isolating the carryover aliquot on the left side (frames 1−4). An example of an open valve can be seen in frame 2, in which B is open and the entire valve is filled with the concentrated dye solution. The mix program is used to mix the diluent and the isolated carryover by serially actuating A, B, and C while I and O are kept closed (frames 5−8). The output reservoir, R O, was manually evacuated in the time between frame 7 and frame 8 for the purpose of visualizing the fully mixed sample. Full movies are included in the.
A flush operation coupled to a mix operation constitutes a microfluidic serial dilution. Sample in the loop can be serially diluted many times to bring about consecutive serial dilutions of the concentrated sample. This concept is presented in.
The detector was positioned between valves A and B (, inset) to observe three consecutive serial dilutions of fluorescein dye concentrate (300 nM). As the dye is cyclically pumped, the concentrated dye signal is acquired. Next, flush(100,60) and mix(100,120) are run sequentially to perform the serial dilution. The measured fluorescence is reduced to background as the buffer diluent passes the detector during flush, then a mixing transient is observed during mix as the diluent and carryover mix. Once mixing is complete, the same program sequence is repeated to generate multiple serial dilutions (, inset). Quantitative evaluation of serial dilution.
(A) Three consecutive serial dilutions of fluorescein dye solution (300 nM in TAE buffer) into TAE buffer were monitored using confocal fluorescence microscopy. The detector position is indicated in the inset circuit schematic. The second and third dilutions are shown in the five-fold magnified inset. Serial dilutions were performed by executing flush(100,60) followed by mix(100,120). (B) A standard curve for 10 μM fluorescein was constructed from the average fluorescence intensity of the sample concentrate, and the intensity obtained after each of four consecutive six-fold dilutions.
Serial Dilution Lab
Each data point represents the average of eight independent experiments. The log plot exhibits excellent linearity over the three detectable orders of magnitude (R 2 = 0.999). The construction of a complete series of standards based on a single 10 μM fluorescein standard solution is presented in.
The log of the fluorescence intensity after each serial dilution was plotted as a function of the serial dilution cycle number, which is expected to be linear with slope proportional to the log of the carryover fraction (CF) of the circuit. The intrinsic CF for a circuit is determined by the fraction of the mixing loop bounded by valves I and O containing valve C.
This fraction linearly depends on the angle θ subtended by the arc between valves I and O (, inset). The CF of circuits with various θ was measured and plotted as a function of θ. Linear agreement of CF with θ is excellent (R 2 = 0.998). The error associated with each CF determination was 1.5%. During flush steps the carryover is still in contact with the flushing diluent stream, so carryover sample near the I and O valve boundaries may diffuse into the diluent stream.
As the diluent flush time is increased, more sample diffuses out and the CF decreases. The dependence of CF on flush time was studied using fluorescein dye and buffer, and found to vary by 5% over the range of 30−300 s. Dependence of carryover fraction on device geometry.
The carryover fraction was related to the arc subtended by valves I and O. The inset indicates the angle measurement, θ. CF = −0.02 + 0.005 θ; R 2 = 0.998. Microfluidic devices are characterized by the reproducibility of operations such as mixing and dilution because the flow regime is laminar. This concept is illustrated in.
Replicate observations of a serial dilution conducted on two different devices demonstrate not only the reproducibility of dilutions performed in the same circuit, but also of dilutions performed on different devices. The inset of presents an overlay of the replicates.
Given identical fluidic programming, the rate of diluent flushing and the oscillations in the mixing transient are reproduced exactly between replicates. Mixing reproducibility. A solution of fluorescein dye was diluted using a circuit with a carryover fraction of 0.12. Two separate devices were operated with identical pumping parameters: flush(100,90), mix(100,120). The five profiles are offset by 200 CPS for clarity.
The start of the flush and mix programs is indicated by arrows. The inset contains an overlay of the five replicates and a sample fit of an exponentially damped sinusoid. Diluent flushing and mixing are highly reproducible, with mixing transients agreeing in fit within 1%. In order to study the reproducibility of the mixing transient quantitatively, a dampened sinusoid was fit to the data. The functional dependence of the damped sinusoid, Ae − kt sin( ω t) + b, contained least-squares fit parameters A, k, ω, and b, corresponding to the amplitude, damping factor, frequency, and offset after dilution, respectively. Typical R 2 values ranged from 0.90 to 0.98.
Parameter ω was fit with less than 0.3% least-squares error, and the frequency determined from fits of the five replicates agreed within 1%. A typical fit curve is shown in the inset, offset from the overlay. Values of R 2 less than 0.95 are attributed to a relatively poor description of damping by the exponential term. Nonetheless, this procedure yielded excellent data on the transient frequency for the purpose of demonstrating the reproducibility of mixing. The time required to mix the diluent plug into the carryover plug is influenced by the pumping rate, or valve actuation time, during cyclic mixing. Presents the dependence of the mixing transient morphology on the valve actuation time.
As the valve actuation time is decreased from 300 ms to 50 ms, the linear flow velocity increases, and the mixing transient is compressed in time. As the two plugs are pumped through each other, mixing is expedited by the establishment of more diffusion planes. The dependence of mixing time on valve actuation time can be determined qualitatively from. At 50 s, for example, the fluorescence intensity is still widely varying in the 300-ms case, while the signal has completely steadied in the 50-ms case.
Mixing transients at variable valve actuation times. (A) Mixing transients were generated with variable actuation times and aligned to time t = 0. (B) Standard deviations as a function of time are plotted as solid lines, sampling valve actuation times of 300, 200, 100, and 50 ms. The standard deviation window width is the period of the oscillation for each transient. A dashed line at σ win = 300 CPS indicates the threshold for complete mixing.
Mixing times (.) measured at different valve actuation times are plotted discretely with respect to the left axis. Mixing times determined by this method exhibited ∼5% standard error.
A quantitative study of mixing time is presented in. The standard deviation of an n-second-wide window, σ win, was plotted as a function of time to measure signal variance. The window width, n, was normalized by setting it equal to the transient period, 2π/ω, determined by fitting a damped sinusoid to each transient (described above). The deviation predictably drops as mixing proceeds. When the carryover and diluent are completely mixed, the standard deviation of the signal is limited by the shot noise of the detector, σ bkgd.
The mixing time is the time required for σ win to reach 2σ bkgd. At this limit of detection, the observer is theoretically unable to differentiate between contributions to signal variance that arise systematically (as a result of incomplete mixing) versus those that arise randomly (as a result of shot noise). An analysis of mixing time as a function of valve actuation time, plotted discretely in, reveals that mixing is expedited as valve actuation time is decreased from 300 ms to 80 ms.
The time required for complete mixing is minimized from 150 s to 22 s over the range of actuation times studied. Further decreasing the valve actuation time from 80 ms to 50 ms did not significantly affect the mixing time.
This agrees with measurements of linear flow rate as a function of valve actuation time; valve actuation appears to be limiting at valve actuation times shorter than 80 ms. The flow rate over the range of 80- to 50-ms valve actuation times gradually becomes independent of valve actuation time. Additionally, at higher flow velocities, transverse diffusion is limiting and the mixing time cannot be decreased absent a mechanism for establishing new boundary layers, for example by promoting torsional flow., Serial dilution is a common operation in chemical measurements. The construction of a series of standard samples can be time consuming and expensive, requiring many fluid metering steps and expending potentially valuable sample. The circuit described here carries out serial dilutions in 400 nL, though this is not a limit for circuit size. In practice this circuit could be scaled down or up depending on the desired sample volume. Design constraints would include the valve dead volume and carryover channel volume.
This microfluidic circuit can generate an entire standard curve with only the diluent as an input. The standards are prepared in nanoliter quantities, conserving reagent and allowing facile integration with on-chip analytical techniques. For example, on-chip capillary electrophoresis or liquid chromatography could be coupled to the output of this circuit, relying on integrated pumping for standard injection. Importantly, this device can execute rapid and automated serial dilutions on the time scale of replication of a population of evolving RNA molecules, opening new avenues of inquiry in molecular evolution. CONCLUSIONS A microfluidic serial dilution circuit was developed that can perform multiple serial dilutions, with greatly increased speed and precision compared to manual pipeting.
Based on alterations of the circuit geometry, carryover fractions of 0.04−0.2 were demonstrated. The circuit requires only 400 nL of starting material to perform an arbitrary number of serial dilutions in a manner that can be integrated with other on-chip preparative or analytical steps. This circuit was developed to enable the automated serial dilution of a population of evolving RNA molecules, but is more generally applicable to almost any microfluidic architecture that involves serial dilution coupled to chemical synthesis or analysis.
Part C: UV Experiments Serial Dilutions and Viable Cell Counts The experiment Observing the Effects of Solar Ultraviolet Radiation on Cells shows that when cells are exposed to sunlight all, some, or none of them may be killed. Many experimental questions can be answered with qualitative answers like 'all, some, or none.' Other questions may require quantitative answers. For example, in the next experiment you will use the sensitive yeast strain to measure the intensity of solar UV radiation by measuring the fraction of cells exposed that survive. To get quantitative answers about yeast survival you must put a known numbers of viable (living) cells onto the agar plates and then count the number that remain after being exposed. You can determine the number of viable cells by counting the colonies that grow up on the agar growth medium in a Petri plate by assuming that each colony grows from a single viable cell. This is usually a reasonable assumption.
Experiment: In the experiment that follows you will learn how to measure the number of viable cells on a Petri plate. You will be able to use this procedure whenever you need to measure the number of cells that survive an exposure to radiation or some other treatment.
First you will estimate the number of cells in a liquid suspension in order to plate a reasonable number of cells. For this you will use one of the most sophisticated and sensitive optical instruments in existence, the human eye.
With surprisingly little practice you can learn to estimate the number of cells in a suspension by just looking at it. You can estimate cell density because of your eyes' fairly sharp threshold for observing turbidity (cloudiness). When viewed in a standard 13 100 mm glass tube, yeast suspensions of less than about 1 million cells per mL are not visibly turbid. Above this threshold density, the suspension is cloudy.
When you adjust the number of cells in a suspension until just barely visible, you obtain a suspension of known density (approximately 1 106 cells/ml). When you have a suspension that contains approximately 1 106 cells/ml, you will dilute it to get the right concentration for plating.
You will make the dilutions in known steps so you can calculate the number of cells in each dilution tube. This procedure helps you plate a countable number of colonies.
There are 2 methods of obtaining pure cultures from samples containing mixed populations. One such method is the dilution and spread plate technique. This method is often used when the mixed populations of prokaryotes exist in a natural sample (i.e. Soil, pond water, fecal material,etc.) and the researcher would like to obtain isolated colonies and enumerate the number of culturable prokaryotic cells in the sample.
The other method of obtaining pure cultures is called streak plate. This method is mostly used to separate mixed populations of prokaryotes in culture (growing culture media). In this experiment, we used serial dilution and spread plate technique to obtain the isolated colonies of bacteria that exists in porridge. Because of their very small size, counting the number of bacteria in porridge can be difficult. Therefore, we used serial dilution and spread plate technique to determine the number of bacteria colony in the porridge. We make a wide range of dilutions of original sample (porridge) to get countable numbers of bacteria in the sample. The porridge is diluted into the diluted factor of 10.
C and 60°C, some doubling in number in as little as 20 minutes. Some types will produce toxins that are not destroyed by cooking. Food poisoning is mainly caused by different and less widespread bacteria.
As they grow, micro- organisms release their own enzymes into the liquid surrounding them, and absorb the products of external digestion. This is the main basis of microbial food spoilage, which lowers its nutritional value. Besides, Bacteria may also produce waste products which act as poisons or toxins, thus causing the renowned ill-effects.
Serial Dilution Lab Conclusion
Since food-poisoning bacteria are often present on many foods, knowing the characteristics of such bacteria is essential to an effective control program. Is found in dust, soil and spices. It can survive normal cooking as a heat-resistant spore, and normal oxygen atmosphere, then produce a large number of cells if the storage temperature is incorrect. Starchy foods such as rice or porridge, macaroni and potato dishes are most often involved. The spores may be present on raw foods, and their ability to survive high cooking temperatures requires that cooked foods be served hot or cooled rapidly to prevent the growth of this bacteria. Therefore, Improper holding and strorage temperatures after cooking, the food will be spoilt and will cause mild case of diarrhea and some nausea within 12 to 24 hours. Escherichia coli Can produce toxins that are heat stable and others that are heat-sensitive.
The major source of this bacteria in the environment is probably the feces of inf ected humans, but there may also be animal reservoirs. Feces and untreated water are the most likely sources for contamination of food. Meat and cheeses are involved the growth of this bacteria.
Windows vista starter iso free download. 5 days ago - Windows Vista Download – Free All Versions ISO DVDs. By Softlay Editor. For 32bit systems only. Free Download Windows Vista Starter ISO.
Serial Dilution Lab Conclusion
It will cause diarrhea and abdominal cramps to consumer. To preventing food poisoning, food handler need to wash their hand and utensils thoroughly before and after hand ling raw foods to prevent recontamination of cooked foods. Food consumer also need to wash hand before and after consume food. Next, refrigerated foods must Keep below 40 degrees F while hot foods must serve immediately or keep them heated above 140 degrees F. Besides, canned foods need to heat thoroughly before tasting.
If a food has been left in the 'Danger Zone'.
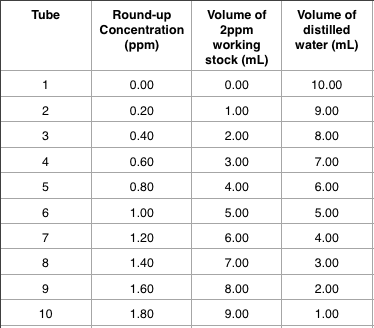
Pre Lab: Serial Dilutions Practice Exercise If students are not familiar with operating pipettes, the Pre Lab Serial Dilutions practice exercise is recommended. The exercise will allow students to learn how to pipette, dilute samples and calculate the concentration of their samples. The use of methylene blue gives them a visual model of how samples become less concentrated with each dilution step. This activity was adapted from An Introduction of Biotechnology developed by the Mathematics and Science Education Center, University of Missouri- St.
A student answer sheet and teacher Power Point are attached for use during this lab. The Pre Lab Exercise can be completed in forty five minutes. Materials Needed Pre Lab Serial Dilutions Equipment and materials for students Equipment and notes Quantity per group (recommend 2-4) □ Test Tube Rack 1 □ Test Tubes 4 □ Flask (recommend 1000mL) 1 □ Pipette Pump (10 mL) 1 □ Pipette (10 mL) 1 □ Methylene Blue (preferably in a dropping bottle) 6 drops □ Water 1 Full Flask □ Lab Tape 2 inches □ Lab Coat or Apron 1 per person □ Safety Glasses 1 per person □ Safety Gloves 1 pair per person □ Calculator (Optional) 1 Preparation Instructions Set up the test tube racks with four test tubes per group. Place the other equipment and supplies at the student work stations or in a central location where they can be located by group members and taken to their work stations. Class Demonstration/Pre-activities If your students are not familiar with how to use a pipette and pump, review the proper way to attach the pipette and pump and how to hold the equipment and how to measure accurately with the pipette. Also, review how to mix the solutions in the tube by using the pipette to draw in the contents of the tube and then releasing back down.
Repeat that step two times for a full mix. Pre Lab Definitions Use the to review the pre lab definitions that the students have on their lab sheet. Serial: In a series, order or interval. Measured steps. Dilution: Water Down.
Pipette: “Little pipe” for moving and measuring liquids. Serial dilutions reduce the concentration of a sample in small steps or fractions. Conducting the Lab The can be used to reveal instructions to the students during the lab. The teacher may wish to go step by step, making sure that all groups have completed a step before moving on to the next one. This will allow the teacher to view the student’s pipetting technique and correct any problems that arise. Students should wear protective aprons or lab coats, gloves and safety glasses due to the use of methylene blue during this lab.
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
